(MENAFN- PR Newswire)
SOUTH SAN FRANCISCO, Calif., Sept. 30, 2024 /PRNewswire/ -- Spiral Therapeutics, Inc. (Spiral), a clinical-stage company focused on developing novel therapies for inner ear disorders, announced the successful completion of the Phase 1b/2a clinical trial of SPT-2101 (6% dexamethasone) in Meniere's disease. Data review of 21 patients showed a significant reduction in vertigo frequency and severity.
The study recruited 21 patients across sites in Perth, Melbourne, and Sydney. The trial included two cohorts:
Continue Reading
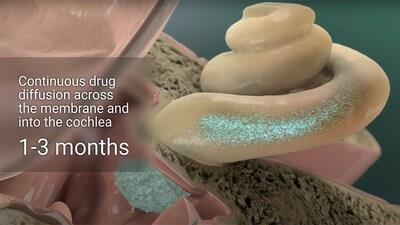
SPT-2101 for Meniere's disease
Cohort 1 : Single administration of SPT-2101 (6% dexamethasone) to 10 patients.
Cohort 2 : A 1:1 randomization with 6 patients receiving a single administration of SPT-2101 (50 μL of 6% dexamethasone) and 5 receiving a control (saline intratympanic injection), with an optional crossover opportunity for non-responders (3 patients crossed over).
Using Spiral's proprietary Minimally Invasive Cochlear System (MICSTM) platform in the clinic, dexamethasone was delivered precisely to the round window membrane for extended release in all study participants. No serious adverse events (SAEs) or unexpected adverse events (AEs) were reported, and no negative impact on hearing was observed. All patients experienced full resolution of the myringotomy.
The treatment with SPT-2101 showed superior vertigo management compared to the control group, with a statistically significant difference (p< 0.05). Additionally, non-responders in the placebo group who crossed over to receive SPT-2101 demonstrated a dramatic reduction in Definitive Vertigo Days at Month 3 (78.7%). The data suggest higher dexamethasone exposures correlated with improved vertigo management across the treatment period, regardless of baseline disease severity.
Dr. Habib Rizk, Director of the Vestibular Program at the Medical University of South Carolina and President of the Board of Directors of the Vestibular Disorders Association (VeDA), commented, "These preliminary results for SPT-2101 are exciting for Meniere's disease patients debilitated by their vertigo episodes. Reliable delivery of steroids into the inner ear may be the key to having patients achieve remission. I am looking forward to seeing Spiral Therapeutics progress with Phase 3 studies to confirm these promising findings."
Dr. Charles Limb, Chief of the Division of Otology, Neurotology, and Skull Base Surgery at UCSF and Chief Medical Officer of Spiral Therapeutics, added, "The encouraging outcomes from this trial reflect Spiral's innovative approach, which combines the concurrent development of a novel therapeutic medication together with a unique delivery procedure using the world's smallest microendoscope for clinical use in the ear. We look forward to continuing our clinical trials so that we can finally offer an effective treatment for Meniere's disease."
The clinical results of SPT-2101 (6% dexamethasone in a crosslinking gel formulation with an estimated two months of drug delivery time) surpass those of Otonomy's OTO-104 (6% dexamethasone in a thermoreversible gel formulation) that was administered with a standard intratympanic injection for the treatment of Meniere's disease and had consistently shown a small efficacy signal on the management of severe vertigo in this patient population. Spiral acquired data on OTO-104 and a right of reference from Otonomy in 2023. Across all measured endpoints, SPT-2101 demonstrated superior outcomes, validating Spiral's approach.
"We are excited to share these positive early results that not only demonstrate the efficacy of SPT-2101 but also reinforce the value of the MICSTM platform, which ensures and extends drug exposure to the inner ear," said Hugo Peris, Chief Executive Officer of Spiral Therapeutics. "These positive results mark a significant milestone in our mission to address the unmet needs of patients with Meniere's disease. The ability to deliver precise, sustained doses of medication directly to the inner ear while remaining minimally invasive is a critical advance for the field."
Spiral's MICSTM platform provides precise and sustained drug delivery directly to the cochlea, addressing a key challenge in therapeutic treatment of the inner ear while maintaining a minimally invasive, clinic-friendly approach. This new paradigm is expected to enable successful clinical translation for a broad range of therapeutics that have shown preclinical promise for treating inner ear disorders.
Data Presentation
Spiral presented these results at a breakfast meeting with key opinion leaders during the American Academy of Otolaryngology Annual Meeting 2024 in Miami, FL, on September 28th. Further analyses are ongoing, and additional results will be reported in the coming weeks.
About SPT-2101
SPT-2101 is a long-acting dexamethasone formulation developed using Spiral Therapeutics' Minimally Invasive Cochlear System (MICSTM) for precise delivery to the round window membrane. This innovative platform ensures targeted, sustained drug release directly to the cochlea, addressing significant unmet needs in inner ear disorders. In clinical trials, SPT-2101 has shown promising results, with a substantial reduction in vertigo for patients with Meniere's disease, significantly improving their quality of life.
About Spiral Therapeutics
Spiral Therapeutics is a clinical-stage biopharmaceutical company dedicated to developing innovative therapies for inner ear disorders. Leveraging its proprietary Minimally Invasive Cochlear System (MICSTM), Spiral enables precise, durable, and minimally invasive drug delivery directly to the cochlea. This groundbreaking platform addresses significant unmet medical needs in treating conditions such as hearing loss and balance disorders. Spiral's therapeutic pipeline includes promising candidates aimed at improving the lives of patients suffering from these challenging conditions. For more information, visit:
.
Media Contact:
Hugo Peris
650-453-0893
[email protected]
SOURCE Spiral Therapeutics, Inc.
WANT YOUR COMPANY'S NEWS FEATURED ON PRNEWSWIRE?
440k+
Newsrooms &
Influencers
9k+
Digital Media
Outlets
270k+
Journalists
Opted In
GET STARTED
MENAFN30092024003732001241ID1108729586
Legal Disclaimer:
MENAFN provides the information “as is” without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the provider above.