
Astrazeneca Withdraws Covid-19 Vaccine Citing Low Demand
AstraZeneca is withdrawing its highly successful coronavirus vaccine, citing the availability of a plethora of new shots that has led to a decline in demand.
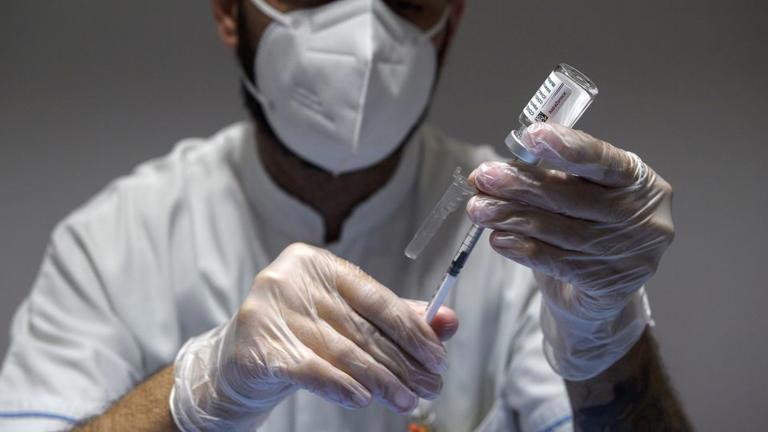
The vaccine - called Vaxzevria and developed in partnership with the University of Oxford - has been one of the main Covid-19 vaccines worldwide, with more than 3 billion doses supplied since the first was administered in the United Kingdom on January 4, 2021.
But the vaccine has not generated revenue for AstraZeneca since April 2023, the company said.
“As multiple, variant Covid-19 vaccines have ... been developed, there is a surplus of available updated vaccines. This has led to a decline in demand for Vaxzevria, which is no longer being manufactured or supplied,” it said in a statement shared with CNN Wednesday.
“AstraZeneca has therefore taken the decision to initiate withdrawal of the marketing authorizations for Vaxzevria within Europe,” it added.
In a notice on its website, the European Medicines Agency also announced the withdrawal, which means that Vaxzevria is no longer authorized to be marketed or sold in European Union countries.
AstraZeneca said it would work with regulators in other countries to“align on a clear path forward,” including withdrawing marketing authorizations for the vaccine where no future commercial demand is expected.
“We are incredibly proud of the role Vaxzevria played... Our efforts have been recognized by governments around the world and are widely regarded as being a critical component of ending the global pandemic,” AstraZeneca said. (CNN)

Legal Disclaimer:
MENAFN provides the
information “as is” without warranty of any kind. We do not accept
any responsibility or liability for the accuracy, content, images,
videos, licenses, completeness, legality, or reliability of the information
contained in this article. If you have any complaints or copyright
issues related to this article, kindly contact the provider above.

















Comments
No comment