
Smith+Nephew's REGENETEN◊ Bioinductive Implant Shows Post-Operative Recovery Times Halved, And High Healing Rates For Partial-Thickness Rotator Cuff Tears1,*
Smith+Nephew (LSE:SN, NYSE:SNN), the global medical technology company, today announces compelling new randomized control trial (RCT) data confirming significant outcome improvements for patients with partial-thickness rotator cuff tears treated with the REGENETEN Bioinductive Implant in isolation.1,*
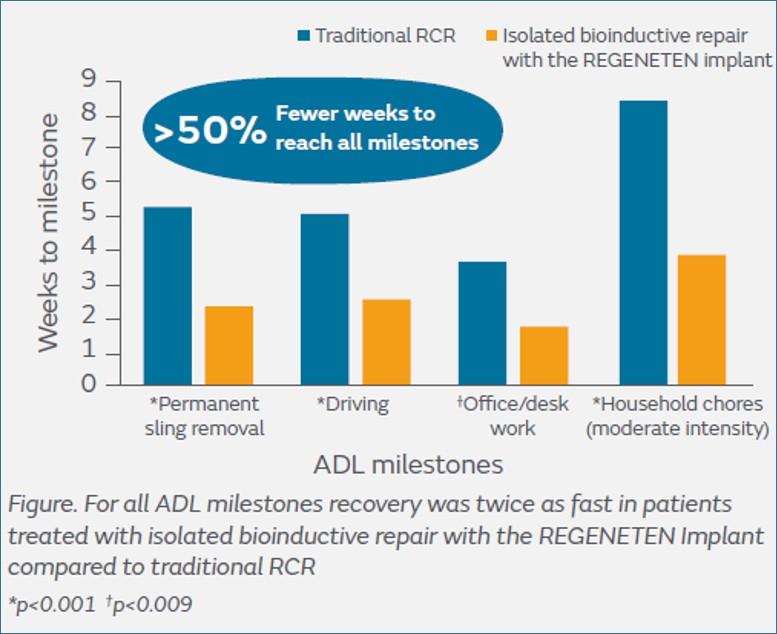
Compared with a traditional suture anchor repair, early recovery time was halved1 for patients returning to daily activities such as:
- Driving: 2.3 vs 5.1 weeks (p<0.001)1,** Office work: 1.7 vs 3.6 weeks (p=0.009)1,** Moderate-intensity household chores: 3.8 vs 8.4 weeks (p<0.001)1,**
The study, recently published in the Orthopaedic Journal of Sports Medicine, also concluded that REGENETEN Implant patients enjoyed a better quality of life at 6 weeks and 3 months, and high rates of tendon healing at 1 year, comparable to suture anchor-based repairs.1 This is the third RCT demonstrating that the REGENETEN Bioinductive Implant improves outcomes versus traditional rotator cuff repair techniques.2-7
“We conducted a randomised controlled study of patients with symptomatic partial-thickness rotator cuff tears and found that those patients treated with the tendon sparing REGENETEN Bioinductive Implant recovered functional independence much faster than patients treated with standard surgery,” said Dr. Allan Wang, Clinical Professor of Orthopedic Surgery at the University of Western Australia.“Time to sling removal, return to driving, office duties, and household tasks all occurred sooner with the REGENETEN Bioinductive Implant. Importantly, these milestones were achieved without an increase in patients' pain and without compromise in rotator cuff tendon healing on 12-month MRI scans. This study marks an important shift in the evolving standard of care for partial-thickness rotator cuff tears.”
◊Trademark of Smith+Nephew. Certain marks registered in US Patent and Trademark Office.

Legal Disclaimer:
MENAFN provides the
information “as is” without warranty of any kind. We do not accept
any responsibility or liability for the accuracy, content, images,
videos, licenses, completeness, legality, or reliability of the information
contained in this article. If you have any complaints or copyright
issues related to this article, kindly contact the provider above.
















Comments
No comment